Medicine and Research of HIV/AIDS
The Identification of the HIV/AIDS virus in the 1980s
 This is Dr. Robert Gallo, the man who discovered the cause of HIV/AIDS.
This is Dr. Robert Gallo, the man who discovered the cause of HIV/AIDS.
In the early 1980s, cases of an unknown disease were popping up across the U.S. It was first thought to be an aggressive form of Kaposi's Sarcoma in young gay men in New York. Also there was an increase in the number of a rare lung infection, Pneumocystis carinii pneumonia, aka PCP, in California and New York [1]. This disease was primarily seen in gay men; by the end of 1981 there was 270 reported cases of this unknown disease and of those 270, 121 of those individuals having already died. By the end of 1981, this disease was then affecting other groups when it was seen in injecting drug users. In the next year, the first clinics for AIDS patients were established and the Center for disease control used the term AIDS for the first time [2]. Within the next years, many scientists rush to find the virus causing this disease and in 1983 Dr. Robert Gallo (shown on the left)[3] suspects that a retrovirus is behind the deadly disease. Gallo and his team, on April 23, 1984, announce that they have found the cause of HIV/AIDS, the retrovirus HTLV-III. This is then followed by the ability to do a blood test to identify the retrovirus in blood samples to stop the spread of AIDS from infected people to uninfected. After the HIV/AIDS cause was found, it immediately became a race to find a vaccine or cure for the disease. By this time, every region of the world had reported a case of AIDS [2].
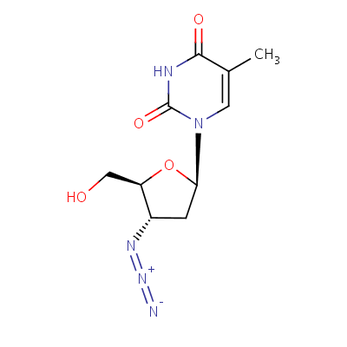 This is the makeup of the medicine Zidovudine.
This is the makeup of the medicine Zidovudine.
In 1987, a medicine was approved by the FDA to help treat HIV/AIDS, called Zidovudine, it is a nucleoside analog reverse-transcriptase inhibitor [2]. This medicine, also known as AZT, is used to treat HIV and reduce the chance of passing HIV from a pregnant woman to her baby. It works by decreasing the amount of HIV in the blood, it does not cure HIV but can stop HIV from developing into AIDS or other serious diseases. This medicine does have harsh side effects such as stomach pain, dizziness, headache, diarrhea, constipation, difficulty sleeping and rash [4]. The medicine works by inhibiting the reverse transcriptase of HTLV-III. It also completely blocks the replication of the virus and the functions of the normal T cells remain intact [5]. The picture on the right is Zidovudine's chemical formula [6]. Also, in 1987, condoms were indicated to prevent the spread of HIV. In 1989, the CDC gives out guidelines to prevent PCP, one of the diseases that caused major illness and even death to many people living with AIDS.
The continuation of the AIDS research into the 1990s
 This is Dr. David Ho on the cover of TIME magazine, he devised the strategy of "hit early, hit hard".
This is Dr. David Ho on the cover of TIME magazine, he devised the strategy of "hit early, hit hard".
In 1990, the FDA approved Zidovudine for use in pediatric AIDS patients. The FDA then in 1992, approves a diagnostic test for HIV that only takes 10 minutes for health care professionals. In 1995, another medicine to treat HIV/AIDS was approved by the FDA, these were protease inhibitors [2]. The first one approved was a medicine called Saquinavir, these protease inhibitors work by attaching to the HIV-1 protease and inhibiting them from replicating but it also works to stop the maturing of already infected cells by inhibiting proteolysis (breakdown of proteins) of the polyprotein within the cells [7]. Many other antiretroviral drugs followed Saquinavir, including: Ritonavir, Indinavir, and Nelfinavir. Finally in 1996 the number of AIDS cases diagnosed decreases for the first time since the epidemic began. The FDA approves easier tests for diagnosis, including: a urine test, a HIV home testing kit and the viral load test, which measures the level of the virus in the person's blood. Also in 1996, Nevirapine is approved, which is the first non-nucleoside reverse transcriptase inhibitor [2]. Nevirapine works in the same way as Saquinavir, in that it inhibits the reverse transcriptase of HIV stopping it from replicating. This drug, however, does not attach itself to the active site of reverse transcriptase but acts noncompetitively [8].
Scientists and Doctors began to use the different drugs that were found to help with AIDS, such as Zidovudine, Nevirapine and Didanosine (another reverse transcriptase inhibitor), together in an antiretroviral drug therapy. Using the drugs together improved the long term effects of therapy, including higher CD4 cell count and lower mean infectious HIV-1 in blood cells. The three drugs together did cause severe rashes to be more common but overall increased the health of the HIV positive patients [9]. Dr. David Ho (pictured on the upper left side of the section) started an initiative to treat patients, "hit early, hit hard", he wanted to treat patients with more aggressive regiment of drugs as soon as they were diagnosed to keep the patients healthier, longer [10]. He was then named TIME magazine's Man of the Year for his work. With this regime, HAART (highly active antiretroviral therapy) becomes the new standard of HIV/AIDS care. In 1997, Combivir was approved by the FDA which was a combination of two antiretroviral drugs in one tablet, this greatly helped people living with HIV to take their medicine. Combivir is a combination of Zidovudine and Lamivudine, it works the same way as those drugs do to stop replication through inhibiting the reverse transcriptase. A dark side to the increase in people getting the drugs they need to treat HIV is that the virus start to become drug resistant to the protease inhibitors [2].
Scientists and Doctors began to use the different drugs that were found to help with AIDS, such as Zidovudine, Nevirapine and Didanosine (another reverse transcriptase inhibitor), together in an antiretroviral drug therapy. Using the drugs together improved the long term effects of therapy, including higher CD4 cell count and lower mean infectious HIV-1 in blood cells. The three drugs together did cause severe rashes to be more common but overall increased the health of the HIV positive patients [9]. Dr. David Ho (pictured on the upper left side of the section) started an initiative to treat patients, "hit early, hit hard", he wanted to treat patients with more aggressive regiment of drugs as soon as they were diagnosed to keep the patients healthier, longer [10]. He was then named TIME magazine's Man of the Year for his work. With this regime, HAART (highly active antiretroviral therapy) becomes the new standard of HIV/AIDS care. In 1997, Combivir was approved by the FDA which was a combination of two antiretroviral drugs in one tablet, this greatly helped people living with HIV to take their medicine. Combivir is a combination of Zidovudine and Lamivudine, it works the same way as those drugs do to stop replication through inhibiting the reverse transcriptase. A dark side to the increase in people getting the drugs they need to treat HIV is that the virus start to become drug resistant to the protease inhibitors [2].
HIV/AIDS research, testing and drugs in the new millennium
 Timothy Ray Brown, first person to be cured of HIV. [11]
Timothy Ray Brown, first person to be cured of HIV. [11]
In the first decade of the new millennium, drug companies began to worry about the state of less developed countries in regards to medicine for HIV positive people. They began to offer reduced drug prices to these countries. In 2002, the FDA approved the first rapid HIV diagnostic test that provided 99.6% accuracy in as quick as 20 minutes. This test was also different because it could be stored at room temperature, could be used outside of a hospital or laboratory and required no special equipment which allowed a greater field for HIV testing. The problem of increasing drug resistance was calling into question the idea of "hit early, hit hard". A San Francisco based company announced in 2003 that their attempt at a AIDS vaccine failed. The testing for HIV continues to improve and in 2004, it is possible to get tested orally instead of through a blood sample and the tests take the same amount of time. In 2006, a study indicated that circumcision of men reduced their risk of getting HIV during heterosexual sex by 53%. This study was supported by the U.S. National Institute for Allergy and Infectious Diseases and by the Canadian Institute of Health Research [2]. In 2007, a man named Timothy Ray Brown, was cured of HIV through his bone marrow transplant. He received two bone marrow transplants, from the same person who was homozygous for the CCR5 protein mutation, this mutation provided resistance in his cells to the HIV virus because they could no longer enter the cells with the mutation. After the transplants, Timothy's levels of HIV dropped significantly and his CD4 T cell count rose. He has been off his antiretroviral drugs since the first transplant and his HIV has not returned since. This cure is not a viable cure for people though, because of it's unlikelihood of working but also the cost. This one cure has pushed research to look into different gene therapy's having to do with the CCR5 allele [12].
HIV/AIDS research and movement towards a cure 2010-2014
In 2010, the National Institutes of Health announce the results of a study which shows that taking a daily dose of HIV drugs reduced the risk of HIV infection in HIV-negative men who have intercourse with HIV-positive men by 44%. Another study was done in 2011 that showed that the risk of HIV infection was reduced for individuals exposed to HIV through heterosexual intercourse [2]. Today, scientists have researched how HIV is becoming drug resistant, through the different mutations that the virus is going through. One such mutation is a mutation on the protease gene, this stops the protease inhibitors, such as Saquinavir, from working correctly. When those drugs aren't working it allows the virus to replicate and mature. There is also mutations in HIV in the reverse transcriptase gene, this causes Zidovudine and other drugs like it, to become obsolete [13]. This drug resistance is becoming more prevalent and could cause a major problem in the near future. Today, there are still many experimental HIV drugs, some are similar to other drugs in their function, like Elvucitabine, which is an experimental nucleoside reverse transcriptase, similar to Zidovudine. There are also new drug treatments like Cenicriviroc, which is an entry inhibitor and there are a whole new category of immune based therapies [14]. The push towards a cure is happening now and with new research and medicine happening constantly, hopefully HIV will be cured not only for one person but for all.
References
[1] "History of AIDS Up to 1986." Avert. Accessed November 19, 2014. http://www.avert.org/history-aids-1986.htm.
[2] "A Timeline of AIDS." AIDS.gov. Accessed November 19, 2014. http://www.aids.gov/hiv-aids-basics/hiv-aids-101/aids-timeline/.
[3] "Robert Gallo." Wikipedia. http://en.wikipedia.org/wiki/Robert_Gallo.
[4] "Zidovudine Oral." MedlinePlus. October 15, 2012. Accessed November 29, 2014. http://www.nlm.nih.gov/medlineplus/druginfo/meds/a687007.html.
[5] Mitsuya, H., and K. Weinhold. "3'-Azido-3'-deoxythymidine (BW A509U): An Antiviral Agent That Inhibits the Infectivity and Cytopathic Effect of Human T-Lymphotropic Virus Type III/Lymphadenopathy-Associated Virus in Vitro." Proceedings of the National Academy of Sciences 82, no. 20 (1985): 7096-100. Accessed November 30, 2014. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC391317/
[6] "Zidovudine." AIDSinfo. May 17, 2014. Accessed November 29, 2014. http://aidsinfo.nih.gov/drugs/4/zidovudine/0/patient.
[7] Mcquade, T., A. Tomasselli, L. Liu, V. Karacostas, B. Moss, T. Sawyer, R. Heinrikson, and W. Tarpley. "A Synthetic HIV-1 Protease Inhibitor with Antiviral Activity Arrests HIV-like Particle Maturation." Science 247, no. 4941 (1990): 454-56. Accessed November 30, 2014. http://www.sciencemag.org/content/247/4941/454.short.
[8] Grob, Peter M., Joe C. Wu, Kenneth A. Cohen, Richard H. Ingraham, Cheng-Kon Shih, Karl D. Hargrave, Tari L. Mctague, and Vincent J. Merluzzi. "Nonnucleoside Inhibitors of HIV-1 Reverse Transcriptase: Nevirapine as a Prototype Drug." AIDS Research and Human Retroviruses 8, no. 2 (1992): 145-52. Accessed November 30, 2014. http://online.liebertpub.com/doi/abs/10.1089/aid.1992.8.145.
[9] D'Aquila, R. "Nevirapine, Zidovudine, and Didanosine Compared with Zidovudine and Didanosine in Patients with HIV-1 Infection. A Randomized, Double-blind, Placebo-controlled Trial. National Institute of Allergy and Infectious Diseases AIDS Clinical Trials Group Protocol 241 Investigators." Annals of Internal Medicine 124, no. 12 (1996): 1019-030. Accessed November 30, 2014. http://europepmc.org/abstract/MED/8633815.
[10] "TIME Magazine Cover: Dr. David Ho, Man of the Year - Dec. 30, 1996." Time. December 30, 1996. Accessed November 30, 2014. http://content.time.com/time/covers/0,16641,19961230,00.html.
[11] "Fred Hutch Hosts Free Community Event with 'Berlin Patient' June 19." Fred Hutch Cure Starts Here. June 7, 2013. Accessed December 10, 2014. http://www.fredhutch.org/en/news/center-news/2013/06/fred-hutch-hosts-free-community-event-with--berlin-patient--june.html.
[12] Hutter, Gero, Daniel Nowak, and Maximilian Mossner. "Long-Term Control of HIV by CCR5 Delta32/Delta32 Stem-Cell Transplantation." The New England Journal of Medicine, no. 360 (2009): 692-98. Accessed December 11, 2014. http://www.nejm.org/doi/full/10.1056/NEJMoa0802905.
[13] Johnson, VA, HF Gunthard, V. Calvez, and R. Paredes. "2011 Update of the Drug Resistance Mutations in HIV-1." Topics in Antiviral Medicine 19, no. 4 (2011): 156-64. Accessed December 10, 2014. http://europepmc.org/abstract/med/22156218.
[14] "New and Experimental HIV Drugs and Treatments." AIDS Action Committee of Massachusetts. December 1, 2014. Accessed December 11, 2014. http://www.aac.org/get-info/health-library-topics/new-and-experimental-hiv.html.
[Banner image] "Access to Medicines." Clinton Health Access Initiative. Accessed December 11, 2014. http://www.clintonhealthaccess.org/program-areas/access-to-medicines.
[2] "A Timeline of AIDS." AIDS.gov. Accessed November 19, 2014. http://www.aids.gov/hiv-aids-basics/hiv-aids-101/aids-timeline/.
[3] "Robert Gallo." Wikipedia. http://en.wikipedia.org/wiki/Robert_Gallo.
[4] "Zidovudine Oral." MedlinePlus. October 15, 2012. Accessed November 29, 2014. http://www.nlm.nih.gov/medlineplus/druginfo/meds/a687007.html.
[5] Mitsuya, H., and K. Weinhold. "3'-Azido-3'-deoxythymidine (BW A509U): An Antiviral Agent That Inhibits the Infectivity and Cytopathic Effect of Human T-Lymphotropic Virus Type III/Lymphadenopathy-Associated Virus in Vitro." Proceedings of the National Academy of Sciences 82, no. 20 (1985): 7096-100. Accessed November 30, 2014. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC391317/
[6] "Zidovudine." AIDSinfo. May 17, 2014. Accessed November 29, 2014. http://aidsinfo.nih.gov/drugs/4/zidovudine/0/patient.
[7] Mcquade, T., A. Tomasselli, L. Liu, V. Karacostas, B. Moss, T. Sawyer, R. Heinrikson, and W. Tarpley. "A Synthetic HIV-1 Protease Inhibitor with Antiviral Activity Arrests HIV-like Particle Maturation." Science 247, no. 4941 (1990): 454-56. Accessed November 30, 2014. http://www.sciencemag.org/content/247/4941/454.short.
[8] Grob, Peter M., Joe C. Wu, Kenneth A. Cohen, Richard H. Ingraham, Cheng-Kon Shih, Karl D. Hargrave, Tari L. Mctague, and Vincent J. Merluzzi. "Nonnucleoside Inhibitors of HIV-1 Reverse Transcriptase: Nevirapine as a Prototype Drug." AIDS Research and Human Retroviruses 8, no. 2 (1992): 145-52. Accessed November 30, 2014. http://online.liebertpub.com/doi/abs/10.1089/aid.1992.8.145.
[9] D'Aquila, R. "Nevirapine, Zidovudine, and Didanosine Compared with Zidovudine and Didanosine in Patients with HIV-1 Infection. A Randomized, Double-blind, Placebo-controlled Trial. National Institute of Allergy and Infectious Diseases AIDS Clinical Trials Group Protocol 241 Investigators." Annals of Internal Medicine 124, no. 12 (1996): 1019-030. Accessed November 30, 2014. http://europepmc.org/abstract/MED/8633815.
[10] "TIME Magazine Cover: Dr. David Ho, Man of the Year - Dec. 30, 1996." Time. December 30, 1996. Accessed November 30, 2014. http://content.time.com/time/covers/0,16641,19961230,00.html.
[11] "Fred Hutch Hosts Free Community Event with 'Berlin Patient' June 19." Fred Hutch Cure Starts Here. June 7, 2013. Accessed December 10, 2014. http://www.fredhutch.org/en/news/center-news/2013/06/fred-hutch-hosts-free-community-event-with--berlin-patient--june.html.
[12] Hutter, Gero, Daniel Nowak, and Maximilian Mossner. "Long-Term Control of HIV by CCR5 Delta32/Delta32 Stem-Cell Transplantation." The New England Journal of Medicine, no. 360 (2009): 692-98. Accessed December 11, 2014. http://www.nejm.org/doi/full/10.1056/NEJMoa0802905.
[13] Johnson, VA, HF Gunthard, V. Calvez, and R. Paredes. "2011 Update of the Drug Resistance Mutations in HIV-1." Topics in Antiviral Medicine 19, no. 4 (2011): 156-64. Accessed December 10, 2014. http://europepmc.org/abstract/med/22156218.
[14] "New and Experimental HIV Drugs and Treatments." AIDS Action Committee of Massachusetts. December 1, 2014. Accessed December 11, 2014. http://www.aac.org/get-info/health-library-topics/new-and-experimental-hiv.html.
[Banner image] "Access to Medicines." Clinton Health Access Initiative. Accessed December 11, 2014. http://www.clintonhealthaccess.org/program-areas/access-to-medicines.
